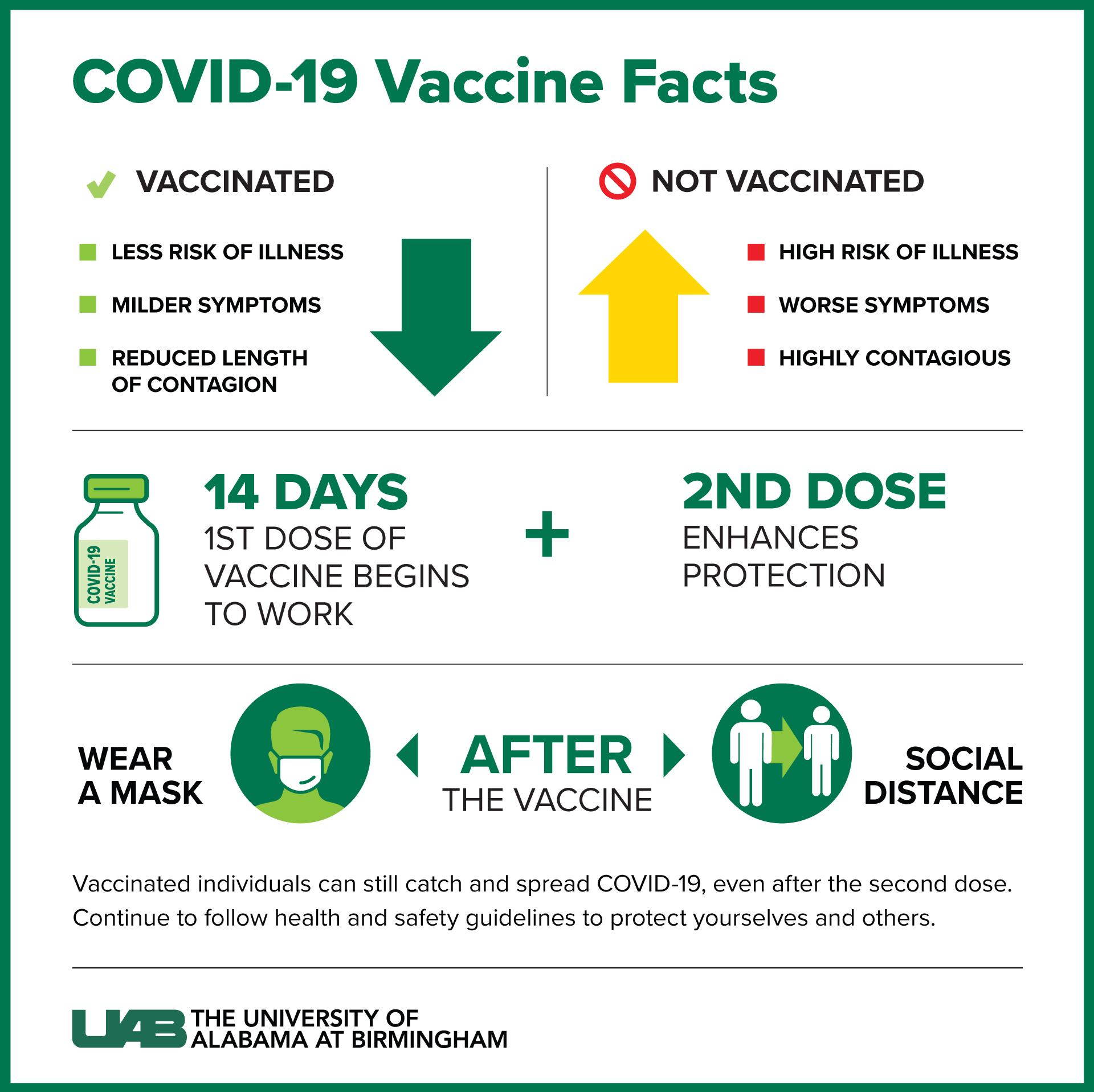
 The vaccine is now also available under an emergency use authorization for children ages 6 months through 4 years old. It also contains a lower amount of mRNA than the Pfizer-BioNTech COVID-19 vaccine used for people age 12 and older. It requires two shots, given three weeks apart. This vaccine is about 91% effective in preventing COVID-19 in children ages 5 through 11. The vaccine is available under an emergency use authorization for children ages 5 through 11. The second dose can be given three to eight weeks after the first dose. For people age 12 and older, the Pfizer-BioNTech COVID-19 vaccine involves two shots. The vaccine is 100% effective in preventing COVID-19 in children ages 12 through 15. The Pfizer-BioNTech COVID-19 vaccine is 91% effective in preventing severe illness with COVID-19 in people age 16 and older. The FDA approved Comirnaty after data found the vaccine is safe and effective. 
The FDA has approved the Pfizer-BioNTech COVID-19 vaccine, now called Comirnaty, to prevent COVID-19 in people age 12 and older. have gotten their booster doses, according to CDC data.Pfizer-BioNTech COVID-19 vaccine. So far, 19% of fully vaccinated people in the U.S. The CDC notes that if any "redness or tenderness" near your injection site gets worse after 24 hours, or if your side effects don't seem to be dissipating after a few days, you should contact a doctor or healthcare provider. The CDC says you shouldn't take these meds before your appointment, because it's unclear whether doing so could interfere with the vaccine effectiveness.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
